Phase Equilibrium
Vapor Pressure
Calculate vapor pressures using Antoine equation. Essential for flash calculations, storage tank design, and RVP blending.
1. Fundamentals
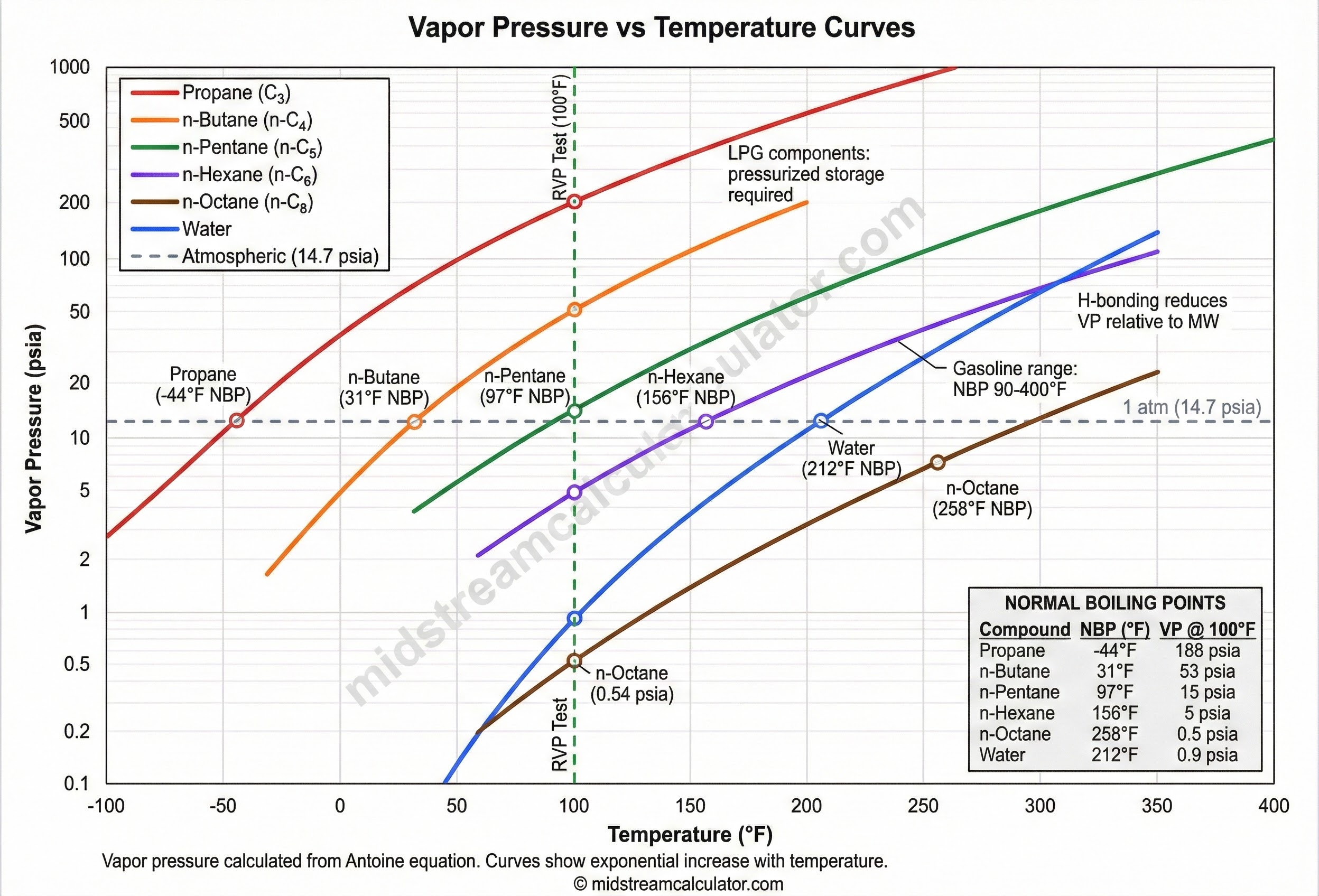
Vapor pressure is the equilibrium pressure exerted by vapor over its liquid at a given temperature. Higher T → more molecules escape → higher VP.
Temperature
Exponential rise
VP roughly doubles every 20°F.
Molecular weight
Lighter = higher VP
Propane > Butane > Octane.
Polarity
H-bonding lowers VP
Water VP lower than MW suggests.
Boiling point
VP = 1 atm
Definition of normal boiling point.
Key Relationships
2. Antoine Equation
Antoine Constants (NIST)
| Compound | A | B | C | T Range (°C) | P units |
|---|---|---|---|---|---|
| Methane | 6.61184 | 389.93 | 266.00 | -181 to -161 | mmHg |
| Ethane | 6.80266 | 656.40 | 256.00 | -142 to -75 | mmHg |
| Propane | 6.82973 | 803.81 | 247.04 | -108 to -25 | mmHg |
| n-Butane | 6.82485 | 935.86 | 238.73 | -78 to 19 | mmHg |
| i-Butane | 6.78866 | 899.81 | 241.94 | -83 to 7 | mmHg |
| n-Pentane | 6.87632 | 1075.82 | 233.36 | -50 to 58 | mmHg |
| n-Hexane | 6.87601 | 1171.17 | 224.41 | -25 to 92 | mmHg |
| n-Heptane | 6.89386 | 1264.37 | 216.64 | -2 to 124 | mmHg |
| n-Octane | 6.91874 | 1351.99 | 209.15 | 19 to 152 | mmHg |
| Water | 8.07131 | 1730.63 | 233.43 | 1 to 100 | mmHg |
| Methanol | 8.08097 | 1582.27 | 239.73 | 15 to 84 | mmHg |
| Ethanol | 8.11220 | 1592.86 | 226.18 | 20 to 93 | mmHg |
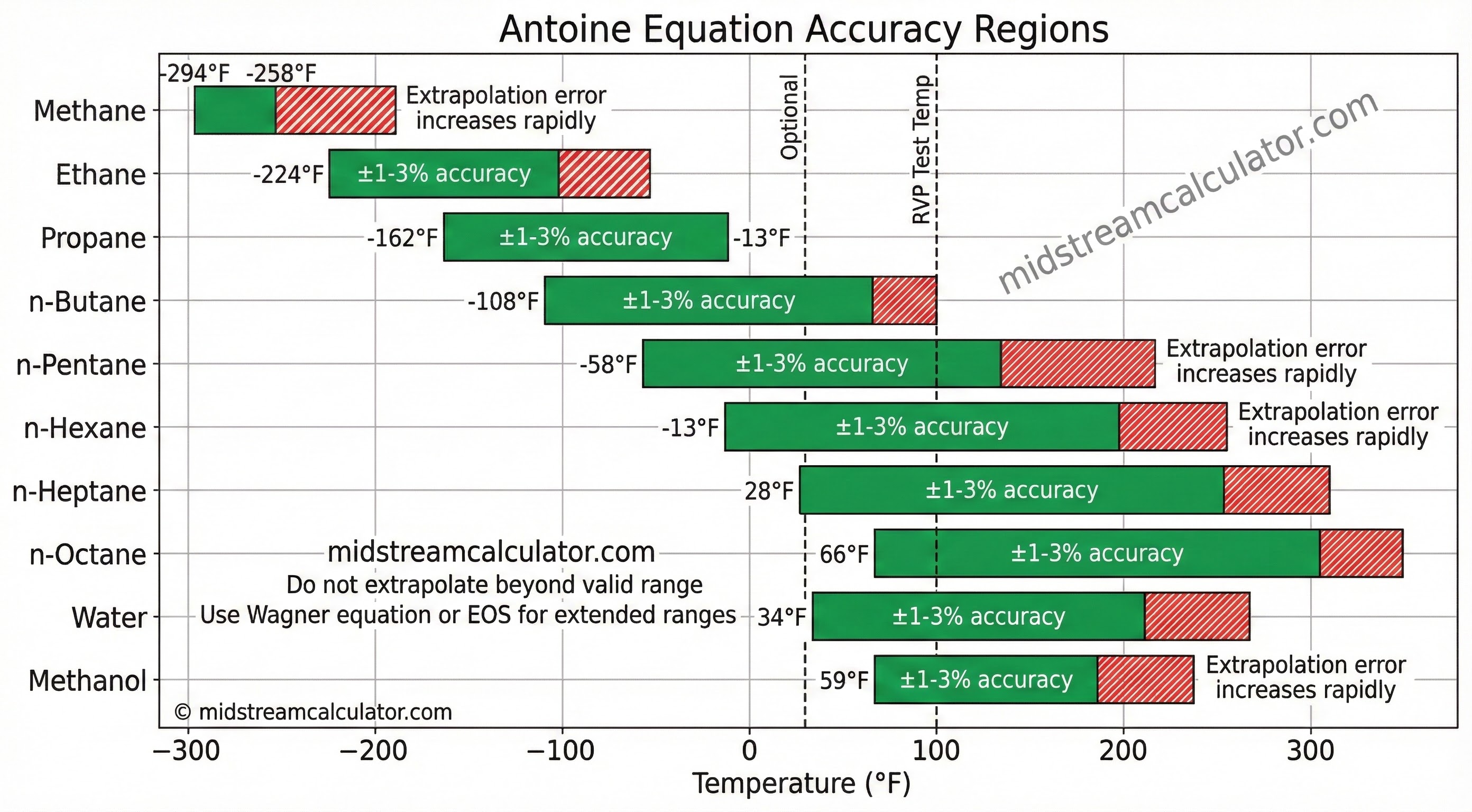
Source: NIST Chemistry WebBook. Always verify T range before use.
Example: n-Butane at 100°F
⚠ Do not extrapolate. Antoine accuracy degrades rapidly outside the valid temperature range. For extended ranges, use Wagner equation or EOS models.
3. Mixtures (Raoult's Law)
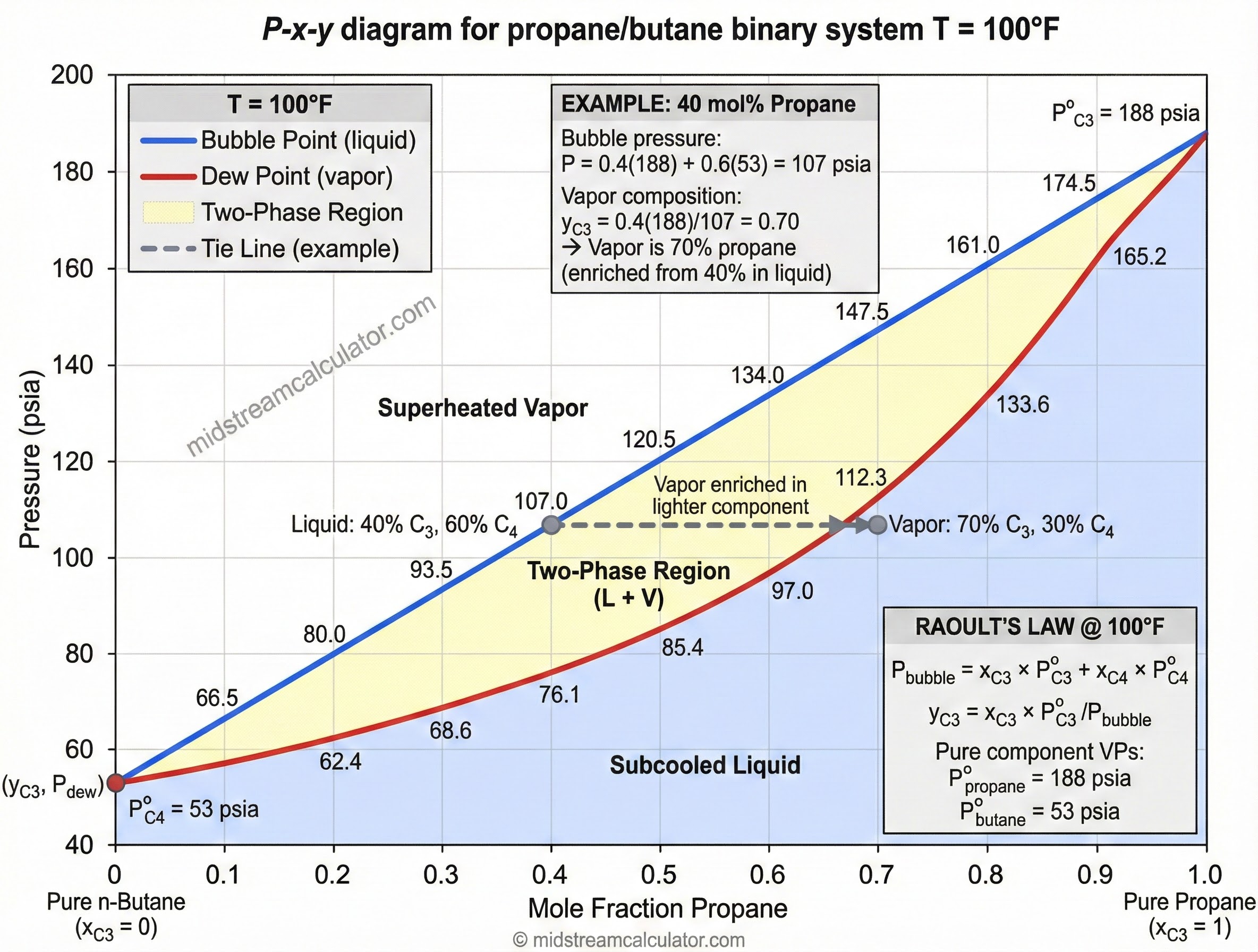
Example: 40% Propane / 60% n-Butane at 100°F
Non-Ideal Mixtures
For polar/nonpolar mixtures, add activity coefficients:
| System Type | γ Value | Recommended Model |
|---|---|---|
| Ideal (similar HCs) | γ ≈ 1.0 | Raoult's Law / Peng-Robinson |
| Slightly non-ideal | γ = 0.8-1.2 | Peng-Robinson with kij |
| Polar/nonpolar | γ = 1.5-5+ | NRTL, UNIQUAC |
| Aqueous alcohols | γ = 2-10+ | NRTL, Wilson |
| Azeotropes | Variable | NRTL with experimental data |
4. Engineering Applications
Flash Calculations
| Parameter | Vapor Pressure Role |
|---|---|
| K-value (ideal) | Kᵢ = Pᵢ°/P_system |
| Bubble point | Σ(xᵢ × Kᵢ) = 1 |
| Dew point | Σ(yᵢ / Kᵢ) = 1 |
| Flash drum sizing | Higher VP → more vapor → larger diameter |
Reid Vapor Pressure (RVP)
| Parameter | RVP | TVP |
|---|---|---|
| Standard | ASTM D323 | ASTM D6378 |
| Temperature | 100°F (fixed) | Actual storage T |
| V/L ratio | 4:1 (fixed) | Actual tank |
| Use | Gasoline spec | Tank venting |
| Typical gasoline | 7-15 psi | Varies with T |
Summer gasoline: RVP 7.0-7.8 psi (EPA limit). Winter: 13.5-15 psi.
Storage Tank Venting (API 2000)
Relief Valve Sizing
Fire relief for liquid-full vessels:
- Step 1: Determine max T from fire heat input (API 521)
- Step 2: Calculate VP at T_max using Antoine
- Step 3: Size PSV for vapor generation at VP
References
- NIST Chemistry WebBook – Antoine constants
- ASTM D323 – Reid Vapor Pressure
- API 2000 – Venting Atmospheric Storage Tanks
- API 521 – Pressure Relief and Depressuring Systems
- Perry's Chemical Engineers' Handbook, Section 2
Ready to calculate?
→ Vapor Pressure Calculator